 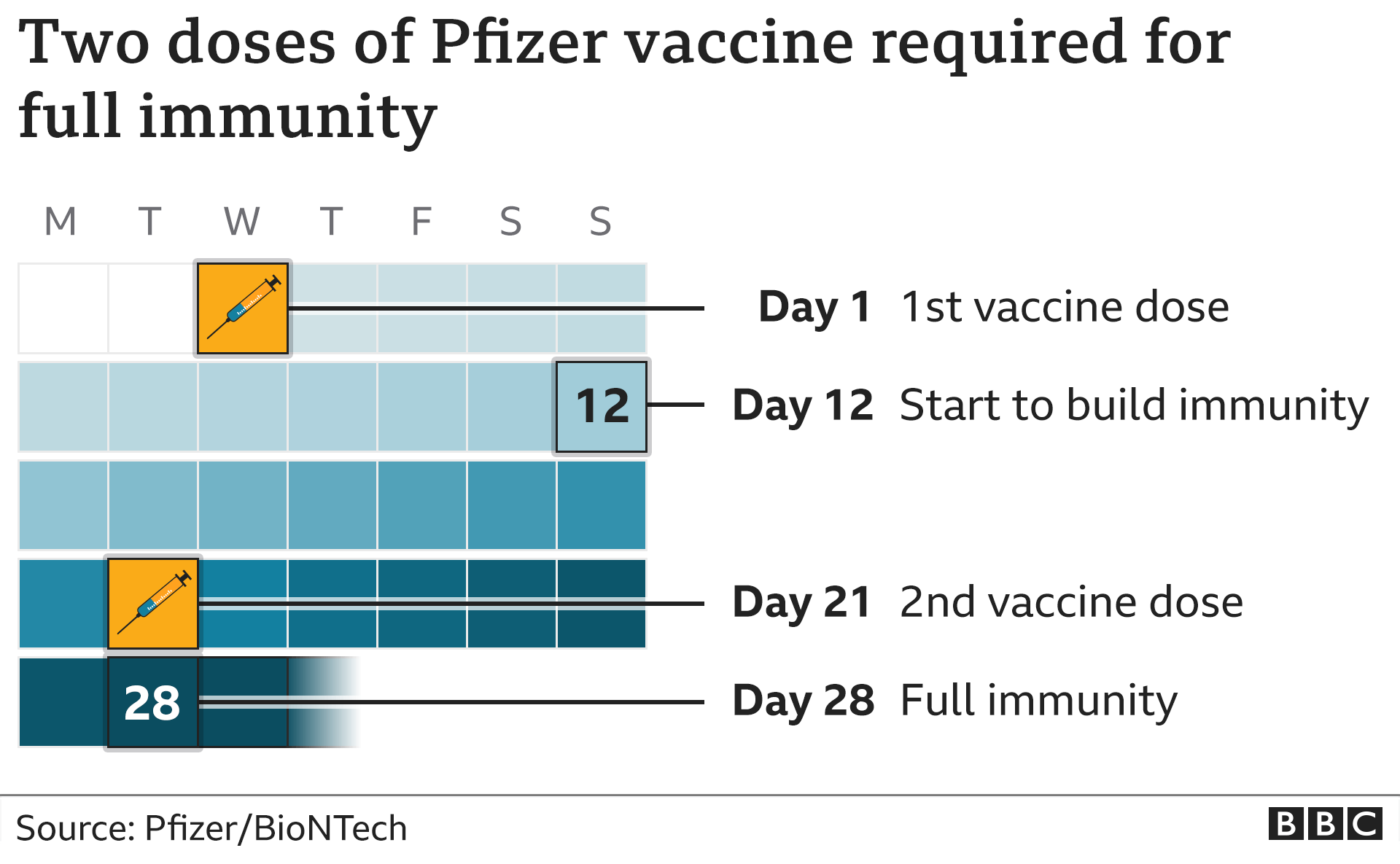
“While absolute risk remains small, the relative risk for myocarditis is higher for males ages 12-39 years, and this risk might be reduced by extending the interval between the first and second dose,” the CDC said, noting that some studies in people older than 12 have shown “the small risk of myocarditis associated with mRNA COVID-19 vaccines might be reduced and peak antibody responses and vaccine effectiveness may be increased with an interval longer than 4 weeks.” Booster doses timingįind out which vaccine types are suitable as boosters.People with Covid-19 may face long-term cardiovascular complications, study says The second dose of this vaccine should be administered at least 3 weeks from your first vaccination. It may be used as a booster for people aged 18 and over if you can't have the Pfizer or Moderna vaccine for medical reasons or if you prefer to have the Novavax vaccine. In Australia, the Novavax vaccine is approved for use in people aged 12 years and over as the primary doses. This ingredient helps to create a stronger immune response to the vaccine. Novavax also has an ingredient called Matrix-M adjuvant. Once you've received the vaccine, your immune system recognises the spike protein as a threat and starts to build an immune response to it. Novavax is a protein-based vaccine, which means it contains part of the COVID-19 spike protein. 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000.Booster vaccinations and primary vaccinations in 6-11 year olds with Moderna are 0.25ml (50 micrograms) Other ingredients One dose (0.5 mL) contains 100 micrograms of messenger RNA (mRNA) (embedded in SM-102 lipid nanoparticles). The ingredients in the Moderna (Spikevax) COVID-19 vaccine are: Active ingredients The second dose of the vaccine should be given 4 weeks after the first dose. booster dose for people aged 16 years and over (50 microgram dose).primary doses for people aged 12 years and older (100 microgram dose).primary doses for people aged 6–11 years (50 microgram dose).In Australia, the Moderna vaccine is approved for use as: The Moderna vaccine doesn’t contain any live virus and it cannot give you COVID-19. Your body will remember and use this immune response if you come into contact with COVID-19. Your body then recognises this spike protein as a threat and triggers an immune response. This means that it doesn’t contain any parts of the virus, but uses a genetic code called RNA to teach your body to make the COVID-19 spike protein. Consumer Medicine Information (CMI) (PDF).2-N,N-ditetradecylacetamide (ALC-0159) Distearoylphosphatidylcholine (DSPC).One dose (0.2 mL) contains 10 micrograms of Comirnaty (tozinameran ) COVID-19 vaccine suspension for injection Other ingredients The ingredients in the Pfizer (Comirnaty) 5–11 year old COVID-19 vaccine formulation are: Active ingredients Therapeutic Goods Administration downloads 2-N,N-ditetradecylacetamide (ALC-0159).((4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate) (ALC-0315).One dose (0.3 mL) contains 30 micrograms of BNT162b2 (embedded in lipid nanoparticles). The ingredients in the Pfizer (Comirnaty) 12 years and over COVID-19 vaccine formulation are: Active ingredients Second booster can be given 3 months after first booster.įind out which vaccine types are suitable as boosters. Booster dose timingįirst booster can be given 3 months after a second dose. Children aged 5–11 should receive their second dose of the Pfizer vaccine 8 weeks after their first dose. People aged 12 and over should receive their second dose of the Pfizer vaccine 3–6 weeks after their first dose. booster doses for people aged 16 years and over (30 microgram dose).primary doses for people aged 12 years and older (30 microgram dose).primary doses for people aged 5–11 years (10 microgram dose).In Australia, the Pfizer vaccine is approved for use as: The Pfizer vaccine doesn’t contain any live virus and it cannot give you COVID-19. You can book to receive your COVID-19 vaccine through a GP or pharmacy. You may have a different brand vaccine for your booster doses. It is important that the first 2 COVID-19 vaccine doses you have are of the same vaccine. There are 5 COVID-19 vaccines provisionally approved for use in Australia of which the following 3 are currently available for use:Įach is approved for specific groups of people and requires a second dose at a specific time. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
